HanAll Biopharma, meanwhile, saw its stock plunge after its partner Immunovant announced that batoclimab failed to meet primary endpoint in its Phase 3 trial for thyroid eye disease(TED). Immunovant added it will focus on a Phase 2 trial of another HanAll asset 'IMVT-1402'.
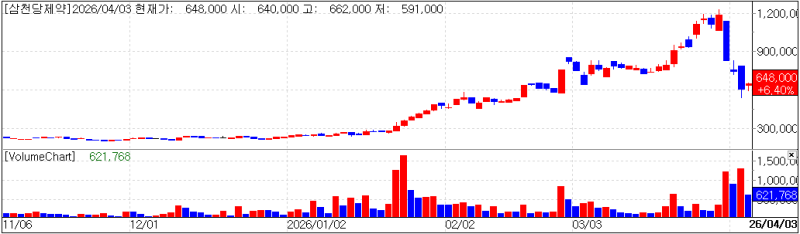
SCD Pharma chart(Cred=KG Zeroin, MP Doctor)
◇Focus shifts to SCD Pharm’s April 6 press briefing
SCD Pharm which once reached a KOSDAQ market cap of 29 trillion won, has been on a rollercoaster falling to fourth place just a week after briefly becoming the top listed company.
The company has promoted its S-PASS technology designed to convert protein drugs into oral pill formulations highlighting prospects such as an “oral obesity drug” and “oral Keytruda.” However criticism has mounted over the lack of evidence backing CEO Jeon In seok’s claim that the technology has been “fully validated.” Doubts have grown as the company has not disclosed patent registrations or study data.
SCD Pharm’s stock had surged after it disclosed licensing deals for oral obesity drug generics in Europe and the United States. Despite Jeon’s plan to sell 250 billion won worth of shares via after hours block trades, investor enthusiasm initially persisted. The stock soared 400% in three months, from 244,500 won at the start of the year to a peak of 1,233,000 won on March 30.
The drop was just as sharp. On the day it peaked, the stock closed at 1,184,000 won then hit the daily lower limit at 829,000 won on March 31 followed by declines to 744,000 won on April 1 and 609,000 won on April 2.
After four days of decline the stock rebounded slightly on April 3, rising 6.4% (39,000 won) to close at 648,000 won. The planned press briefing appears to have restored some investor confidence.
Previously, SCD Pharm had relied on pop-up notices on its official website to respond to allegations. The site currently displays six pop-ups rebutting claims from bloggers, analysts, and media outlets. The company insists that its conditional approval-based contract for oral GLP-1 with an undisclosed U.S. firm under which it would receive 90% of profits is factual.
Industry sentiment remains skeptical. “This controversy could be resolved if the company disclosed S-PASS technology, but it hasn’t, which raises questions,” one industry source said.
SCD Pharm said the upcoming press briefing will outline key growth projects and its future strategy including development of oral diabetes drug Rybelsus generics, development of oral obesity drug Wegovy generics, exclusive licensing and commercialization deals across 11 European countries entry of an oral insulin candidate into European clinical trials, a U.S. exclusive licensing agreement
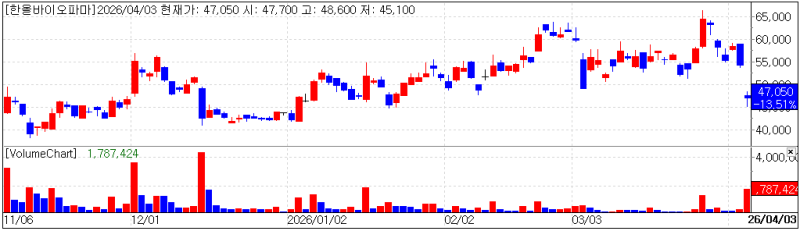
HanAll Biopharma chart(Cred=KG Zeroin, MP Doctor)
◇HanAll Biopharma hit by batoclimab Phase 3 setback
HanAll Biopharma, a new drug developer 30%-owned by Daewoong Pharmaceutical, fell 13.51% (7,350 won) to close at 47,050 won, following news that Immunovant failed in a global Phase 3 trial of batoclimab, an antibody therapy for autoimmune diseases.
HanAll had license-out batoclimab (HL161BKN) and imeropruvate (HL161ANS/IMVT-1402) to Switzerland-based Roivant Sciences in 2017. Roivant later spun off Immunovant to accelerate global clinical development. The pipeline had built expectations over nine years, making the Phase 3 failure a major blow to investor sentiment.
The failed trial targeted thyroid eye disease, which causes proptosis, or eye protrusion. The primary endpoint was a 2 mm improvement at Week 24, which was not met. No new safety issues were identified.
Immunovant said it will discuss future development plans for batoclimab with HanAll. Despite the setback, Immunovant noted that higher-dose treatment during the first 12 weeks showed significantly better improvement than the subsequent lower-dose phase, suggesting efficacy is linked to IgG suppression.
The company added it will continue Phase 2 development of imeropruvate, another FcRn-targeting asset, for Graves’ disease, with topline data expected in 2027.
The total deal size between HanAll and Roivant stands at $502.5 million, including a $30 million upfront payment, $20 million in R&D funding, and up to $452.5 million in milestones, plus royalties on sales. Total payments received by HanAll have not been disclosed.
GI Innovation chart(Cred=KG Zeroin, MP Doctor)
◇GI Innovation gains on ASCO oral presentation selection
GI Innovation rose 7.92% (920 won) to close at 12,530 won. The company said its Phase 1 data for immuno-oncology drug GI-101A has been selected for a rapid oral abstract presentation at ASCO 2026, boosting investor sentiment.
ASCO is one of the most prestigious conferences in oncology, where cutting-edge clinical data are first unveiled and evaluated based on real patient outcomes.
ASCO 2026 will be held in Chicago from May 29 to June 3. GI Innovation will present its Phase 1 results, with the oral session reserved for top-tier research among thousands of submissions.
CEO Jang Myung ho said “The ASCO oral presentation validates the clinical potential and scientific value of GI-101A. We will clearly present its clinical significance and strategic value in combination therapies.”