Kyungnam Pharm hit the daily upper limit after announcing a share consolidation plan aimed at stabilizing its stock price and enhancing corporate value.
VigenCell’s share price climbed as the company decided to terminate early its clinical trial for an acute myeloid leukemia treatment and instead focus its resources on its core pipeline candidate for a rare lymphoma therapy.
Vivozon Pharmaceutical also saw its stock rise on expectations surrounding the full scale commercialization of its globally first-in-class multi-receptor targeted non opioid analgesic Opiranserin marketed under the brand name Unafra, as well as news that its management would actively participate in a rights offering.
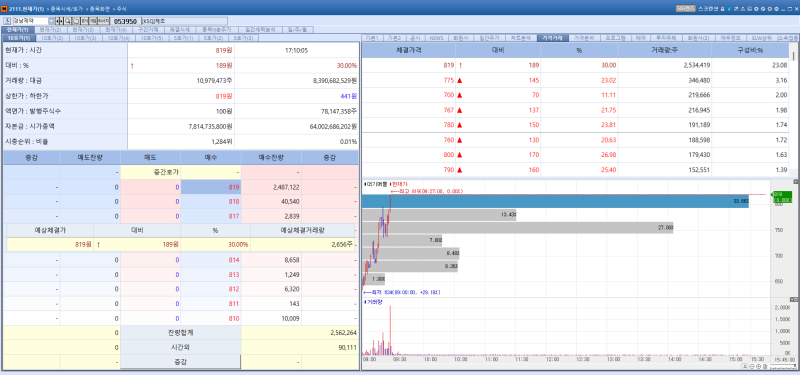
Kyungnam Pharm stock trend on Feb 25.(Image=MP Doctor)
◇Kyungnam Pharm to Consolidate Shares from 100 Won to 500 Won
Kyungnam Pharm’s shares surged 30.00% from the previous trading day to close at 819 won, hitting the daily upper limit. The sharp rise followed the company’s announcement that it would proceed with a share consolidation.
The company will consolidate its common shares with a par value of 100 won into shares with a par value of 500 won. Following the consolidation, the total number of outstanding common shares will decrease from 78,147,358 to 15,629,471. The newly issued shares are scheduled to be listed on May 7. Trading of Kyungnam Pharm’s shares will be suspended from April 14 to May 6.
A share consolidation reduces the number of outstanding shares while increasing the price per share. However, it does not change the company’s overall corporate value. Companies typically opt for share consolidation to meet listing requirements or to attract institutional investors. As the share price is adjusted in line with the consolidation ratio, there may be short term upward movement in the stock.
Kyungnam Pharm has posted operating losses for five consecutive years, reflecting sluggish performance. The company reported 55.9 billion won in sales and an operating loss of 1.6 billion won last year. Revenue declined 8% year on year while the operating loss widened 74.9%.
Founded in 1953 Kyungnam Pharm experienced frequent changes in management control after its founder stepped down in the 2000s. In 2024 Humasis became the company’s new controlling shareholder.
A company official said “We decided to implement the share consolidation to stabilize the stock price and enhance corporate value.”
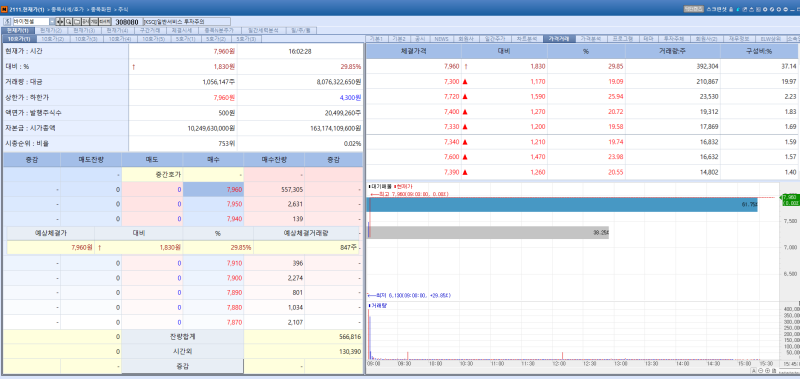
VigenCell stock trend on Feb 25.(Image=MP Doctor)
◇VigenCell Focuses on Rare Lymphoma Drug That Demonstrated Efficacy in Phase 2 Trial
VigenCell’s shares soared 29.85% from the previous trading day to close at 7,960 won. The surge is attributed to the company’s decision to terminate early its clinical trial for an acute leukemia treatment candidate and concentrate resources on its core pipeline programs.
The company announced that it would discontinue the clinical trial of its acute myeloid leukemia (AML) treatment candidate, VT-Tri(1)-A. VigenCell explained that the decision was a strategic move aimed at enhancing shareholder value through the efficient allocation of limited resources. Since enrolling its first patient in 2022, the company had been conducting a Phase 1 cohort 3 study of the AML therapy.
No serious adverse drug reactions (ADRs) were observed in cohorts 1 and 2. However, during cohort 3, one case of Grade 3 graft-versus-host disease (GvHD) was reported.
VigenCell stated that, unlike VT-EBV-N—which is derived from a patient’s own blood—the AML candidate uses blood from a hematopoietic stem cell donor, and the adverse event is presumed to be related to this product characteristic.
The company plans to redirect the resources saved from the early termination toward its rare lymphoma treatment VT-EBV-N and its next-generation growth engine, an induced pluripotent stem cell (iPSC)-derived CAR-NK cell therapy.
Previously VigenCell demonstrated the efficacy of VT-EBV-N in a Phase 2 trial compared with a control group. In the primary endpoint analysis of two-year disease-free survival (DFS), the VT-EBV-N treatment group recorded a rate of 95.00%.
A company official described the result as “an outcome that nearly perfectly prevented patient relapse.”
Moreover no deaths occurred in the treatment group during the clinical trial period, and no notable adverse events were reported, supporting the therapy’s safety profile. In contrast, the control group showed a two-year disease-free survival rate of 77.58% with multiple deaths reported marking a clear difference from the VT-EBV-N group.
Based on the Phase 2 results, VigenCell plans to pursue commercialization of VT-EBV-N. The company aims to seek conditional marketing approval in the second half of this year.
Another company official stated “The results not only secured statistical significance but also demonstrated high practical value in real-world clinical settings.”
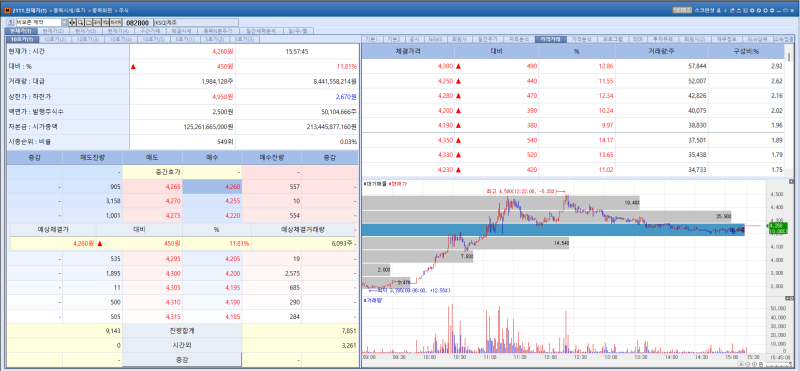
Vivozon Pharmaceutical stock trend on Feb 25.(Image=MP Doctor)
◇Vivozon Pharmaceutical Executives Oversubscribe Rights Offering, Signaling Commitment to Responsible Management
Vivozon Pharmaceutical’s shares rose 11.81% from the previous trading day to close at 4,260 won. The gain is widely attributed to the active participation of the company’s management in the ongoing rights offering, a move interpreted as demonstrating strong commitment to responsible management.
CEO Jang Boo hwan subscribed to 11,000 shares, equivalent to 120% of the shares allocated to him under the offering. Lee Doo hyun Chairman of Vivozon Group also applied for approximately 17,000 shares the maximum number available to him. The oversubscription by top executives is viewed as a clear signal of accountability and confidence in the company’s future.
Vivozon Pharmaceutical is conducting a rights offering through a shareholder allocation followed by a public offering of unsubscribed shares. After finalizing the offering price at 3,065 won on the 19th, the company carried out subscriptions for existing shareholders from the following day through the 24th.
Proceeds from the capital increase will be used for research and development, general operating expenses and debt repayment to improve the company’s financial structure.
Since November last year Vivozon Pharmaceutical has begun full scale domestic sales of its non opioid analgesic Unafra. The product is being co marketed in Korea by Daiichi Sankyo Korea and Hanmi Pharmaceutical.
Unafra, designated as Korea’s 38th domestically developed new drug, is the world’s first multi-receptor targeted non opioid analgesic. The market has placed high value on its rarity as a potential game changing therapy in the global pain management sector, where few breakthrough drugs have emerged over the past decades.
Industry observers project that supported by the sales synergy of major Japanese and Korean pharmaceutical companies Unafra could generate more than 30 billion won in domestic sales. Vivozon is also pursuing global out licensing opportunities alongside its domestic commercialization efforts.
One of Unafra’s key strengths is its ability to treat moderate to severe pain. The domestic market for non opioid analgesics is estimated at approximately 120 billion won.
A company official stated “As domestic sales of Unafra increase, the overall non-opioid analgesics market is expected to expand as well. Amid growing demand in clinical settings for non-opioid pain treatments we aim to lead the market by providing a safe and effective therapeutic option.”










